Electronic nicotine delivery systems, colloquially referred to as e-cigarettes (ECs), or vaping products were introduced in the US market in 2007 as an alternative to cigarette smoking. They were marketed as a better alternative to nicotine replacement therapy for stopping cigarette smoking, as ECs are considered less harmful than tobacco cigarette smoking.1 These findings have been validated in a randomized controlled trial that found significantly better abstinence rates with ECs compared with nicotine replacement (18.0% versus 9.9%).2 A large meta-analysis found conclusive evidence that completely substituting combustible tobacco cigarettes for ECs reduces users’ exposure to numerous toxicants and carcinogens.3 Various health issues have been reported secondary to the use of ECs including thermal injuries, cardiovascular effects and several types of lung injuries, which are outlined in Table 1.4–17

Cases of EC or vaping product-associated lung injury (EVALI), though sporadically reported, have been rising since 2019. In the last updated statement by the Centers for Disease Control and Prevention (CDC), there have been over 2,800 cases reported across the USA, following which these cases were not reported further by the CDC.18 Of note, a retrospective analysis of a large national database in Korea evaluating the association between EC use and severe pneumonia failed to show any cases of EVALI.19 With the coronavirus disease 2019 (COVID-19) pandemic and the significant decrease in the number of cases of EVALI, further reporting of EVALI cases was discontinued by the CDC.18 However, these cases still occur, albeit rarely as there is more control of the adulterated vaping products within the USA. Moreover, the COVID-19 pandemic may be associated with an increased incidence of EVALI due to social distancing and isolation precautions resulting in increased tendency to use ECs.20,21 In this review we will discuss the pathophysiology of EVALI, along with differences between EVALI and COVID-19, and the impact of COVID-19 on EVALI.
Structure of e-cigarettes
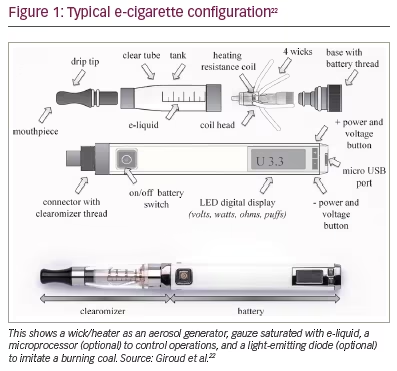
An EC is composed of an aerosol generator, a flow sensor, a battery, a solution (or e-liquid) storage area and a light-emitting diode. The aerosol generator consists of a metallic or ceramic heating-element coil (Figure 1).22 On drawing up the EC, the airflow sensor detects pressure change and activates the light-emitting diode and metallic coil. The e-liquid saturates a wick via capillary action and gets aerosolized by the heating element; the aerosol is subsequently inhaled.23
ECs were originally designed to resemble traditional cigarettes for appeal, but they have evolved over the years. Initially, first-generation ECs were called ‘cig-a-likes’, resembled conventional cigarettes and were usually disposable.24 The second-generation EC (‘tanks’) came with a rechargeable battery and a refillable cartridge, in the shape of a pen (Figure 2).25 Following this, the third-generation ECs (‘mods’) were released, which were bigger and allowed the user to control temperature and heating.26 Most recently, the fourth-generation ECs (‘pods’) have come to market, with rechargeable batteries and refillable pods that come in various flavours.24 These pod-based devices (e.g. Juul [Juul Labs Inc, San Francisco, CA, USA]) have become extremely popular with teenagers and school-going children.26

Epidemiology of e-cigarette or vaping product-associated lung injury
As of 18 February 2020, there have been 2,807 hospitalized EVALI cases or deaths reported to the CDC from the USA, with 68 reported deaths. Among the 2,807 reported cases, 76% were 34 years old or younger, 66% were male, 82% used tetrahydrocannabinol (THC)-containing products and 33% reported exclusive use of THC-containing products.18 Most cases were in teenage and young adult males. However, following the increased incidence of EVALI, the number of people using ECs has decreased; in US high school students, use dropped from 27.5% in 2019 to 19.6% in 2020,27,28 with concurrent decline in the number of EVALI cases as well.18 This can be attributed to public awareness, removal of vitamin E acetate from some products and law enforcement actions related to illicit products.29
Pathogenesis and pathology of e-cigarette or vaping product-associated lung injury
The specific pathogenic mechanisms for EVALI are yet to be completely understood. Lipid-laden macrophages with vacuolization and vacuolated pneumocytes is a common pathological feature seen across multiple EVALI cases;6 this is consistent with chemical-induced pneumonitis.30 In a recent study, 48 out of 51 cases of EVALI (94%) had vitamin E acetate in their bronchoalveolar lavage (BAL) samples.31 Furthermore, THC products were detected in the BAL sample of 47 of 50 cases (94%).32 Vitamin E acetate has been identified as a common culprit;31 however, pathogenesis may be more complicated given the heterogeneity of the products available. EC vaping oils have been associated with seven toxic compounds, including nicotine, volatile organic compounds, carbonyls, small dust particles, metal elements depending on flavours, bacterial endotoxins and fungal glucans.32,33
Volatile organic compounds have long been associated with various ill effects.34 Two common flavouring chemicals used with ECs, diacetyl and 2,3-pentanedione, induce transcriptional changes and downregulate expression of genes, causing ciliary dysfunction at a cellular level and predisposing patients to severe respiratory diseases.34 Aldehydes used in liquid flavouring agents, such as diacetyl and acetylpropionyl, can cause bronchiolitis.35 Glycerin and propylene glycol mixed within the solvent decompose at high temperatures, generating carbonyl compounds – namely formaldehydes, acetaldehyde and acrolein – injuring the airway epithelium through oxidative stress and the inflammatory cascade.4,36 The severe inflammatory response and oedema may result from pyrolysis of products of vitamin E oil (gases such as ketene).31
EVALI is associated with several patterns of lung injury, including fibrinous pneumonitis, diffuse alveolar damage (DAD) and organizing pneumonia (OP), which is usually bronchiolocentric with bronchiolitis. In one study, foamy macrophages and pneumocyte vacuolization was seen in all cases, bringing into question the significance of routine oil red O staining in BAL as a marker for lung injury. This suggests that EVALI is more of an airway-centred chemical pneumonitis rather than exogenous lipoid pneumonia.6 Other lung disease patterns associated with EVALI include diffuse alveolar haemorrhage,16 acute eosinophilic pneumonia, acute respiratory distress syndrome (ARDS), hypersensitivity pneumonitis, fibrinous and peribronchiolar granulomatous pneumonitis, and in rare cases giant-cell interstitial pneumonitis (Table 1).5
Clinical signs and symptoms of e-cigarette or vaping product-associated lung injury
The clinical presentation of patients with EVALI, based on the first described cohort of EVALI patients and subsequently shown in most of the later published studies, is predominantly young males (median age of 21 years) primarily with respiratory symptoms (97%) – such as shortness of breath, cough, chest pain and haemoptysis – and gastrointestinal symptoms (77%), including nausea, vomiting, diarrhoea and abdominal pain.9 In one study, approximately 89% of patients reported using THC-containing products in their e-cigarette devices, although several other products were also used. Of note, progression to hypoxaemic respiratory failure was not uncommon, with at least 65–70% of patients admitted to intensive care units for high-flow oxygen therapy or mechanical ventilation, which may be required in up to 15–30% of patients.5 Factors predicting mortality include non-Hispanic white ethnicity, older age, history of bronchial asthma, cardiac disease and obesity.37
Radiology
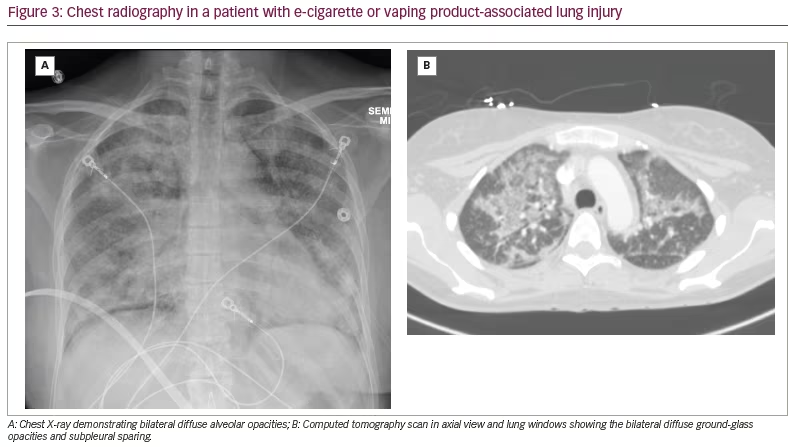
Imaging findings in EVALI are usually consistent with those of toxic inhalational acute lung injury.38 Given that the more common pathological patterns of EVALI comprise OP or DAD, radiological patterns commonly associated with EVALI reflect these pathological patterns.38 When associated with predominantly OP, chest imaging is characterized by diffuse hazy opacities centrally with diffuse ground-glass opacities with subpleural and peri-bronchovascular sparing seen both centrally and peripherally (Figure 3A and B).38 Reflecting the airway-centric injury, centrilobular nodules are another common pattern described in EVALI, which may be secondary to the development of OP foci within the airways.38 When DAD is the predominant pathology, computed tomography (CT) findings include lower-lobe predominant consolidation, which may progress to fibrotic changes with reticulation and bronchial dilatation.39 Other rare findings include interlobular septal and bronchial wall thickening, mediastinal lymphadenopathy, and rare small pleural and pericardial effusions.40–42 In some cases, depending on the intensity of respiratory effort, pneumothorax and pneumomediastinum have been observed.9
Diagnosis
No uniform diagnostic criteria exist for EVALI and it remains primarily a diagnosis of exclusion. The original case definition from the Wisconsin report (first case series of 53 patients with EVALI) included the following:9
1. Use of an e-cigarette (vaping) or dabbing in the 90 days before symptom onset
2. Pulmonary infiltrates, such as opacities on plain-film radiograph of the chest or ground-glass opacities on chest CT
3. Absence of pulmonary infection on initial work-up: negative respiratory viral panel; influenza polymerase chain reaction; urine antigen testing for Streptococcus pneumoniae and legionella; sputum culture if productive cough; BAL culture if done; blood culture; and presence of HIV-related opportunistic respiratory infections if appropriate
4. No evidence in medical record of alternative plausible diagnoses (e.g. cardiac, rheumatological or neoplastic process).26
A urine drug screen for THC, although non-specific, may help identify EC use and aid further diagnosis. No laboratory markers are diagnostic for EVALI. Inflammatory markers including C-reactive protein and erythrocyte sedimentation rate are usually elevated but are non-specific. Bronchoscopy with BAL is helpful to exclude infectious processes or diagnose other associated conditions including diffuse alveolar haemorrhage, hypersensitivity pneumonitis or acute eosinophilic pneumonia. Presence of lipid-laden macrophages in BAL has poor specificity and cannot be suggested as a laboratory marker for EVALI.6,31,43,44 Presence of vitamin E acetate in BAL fluid may be useful.31
Management
The first step in treatment is stopping all forms of smoking. Empirical antibiotics should be considered, as community-acquired pneumonia is much more common. Treatment options for EVALI include corticosteroids and oxygen therapy. Around 95% of patients with EVALI may require hospitalization.9 Subsequently, if hypoxaemia worsens, patients can be transferred to the intensive care unit and treated for ARDS.45
Systemic corticosteroids have been used extensively in patients with EVALI, but data are largely from case series.9,46,47 In a case series of 98 patients, 84% of patients received corticosteroids, though 51% improved.9 In another small case series, intravenous methylprednisone ranging from 120 mg to 500 mg was administered daily; all patients improved within 24–72 hours of treatment.48 Corticosteroids should be initiated in patients who meet the criteria for EVALI.5 An initial dose of 0.5–1 mg/kg with a taper after 7–10 days may be appropriate, but studies are needed to develop an appropriate regimen.5 Response to corticosteroids may vary due to the different pathological injuries caused by EVALI; patients with an OP pattern of injury, eosinophilic pneumonia or hypersensitivity pneumonitis will likely respond to corticosteroids, such as prednisone or methylprednisolone, while patients with DAD or fibrinous pneumonitis are unlikely to respond to corticosteroids.5
Hospitalized patients should be seen in the clinic 1–2 weeks after discharge. Additional follow-up testing, such as pulmonary function tests (PFTs) (spirometry, diffusion capacity) and repeat imaging, should be considered on a case-by-case basis.47 Outpatient management of EVALI should be individualized, and considered in patients with an oxygen saturation >95% and reliable access to healthcare with strong support systems.47
Overall, the prognosis in young patients is excellent, though the long-term consequences of EVALI are still not clear.5 However, short-term follow-ups have either shown persistent clinical, radiographical or PFT abnormalities,49 or a complete clinical and radiographical clearance within 2 weeks of follow-up.50 In a review of 114 patients with EVALI, 31 patients had PFTs performed at 30 days post-discharge; 52% (16/31) of these patients had abnormal PFTs, with the most common abnormalities being obstruction, reduced diffusing capacity for carbon monoxide, or both.51
Coronavirus disease 2019 and e-cigarette or vaping product-associated lung injury
Since the start of the COVID-19 pandemic, diagnosing EVALI has been challenging. A recent observational study showed a dominant negative sentiment regarding vaping/ECs, which was persistent through the pandemic.52 An online survey of adolescents and young adults aged 13–24 years showed that COVID-19 diagnosis was five times more likely in patients who smoke ECs daily, and seven times more likely in patients who smoke both ECs and traditional cigarettes.21
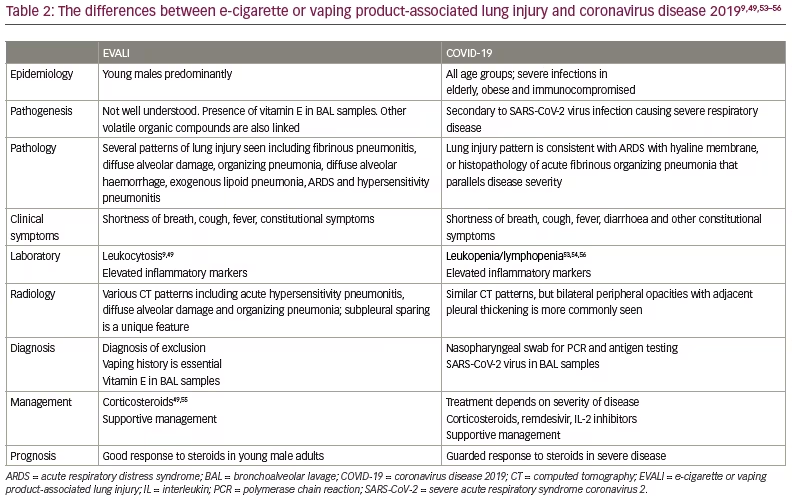
The overlap of clinical and radiological features between the two conditions makes diagnosis of EVALI difficult (Table 2).9,49,53–56 Given the variability in the availability of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) testing, alongside the non-optimal sensitivity and specificity of these tests, and barriers related to aerosolization during bronchoscopy, it is challenging to distinguish EVALI from COVID-19.56 However, subtle differences still exist between COVID-19 and EVALI. Both COVID-19 and EVALI can present with CT findings consistent with acute hypersensitivity pneumonitis, DAD and OP; however, subpleural sparing is more common in EVALI.57 Laboratory data show that COVID-19 may be associated with normal or low white blood cell counts often with lymphopenia, while EVALI is always characterized by leukocytosis.56 Finally, while corticosteroids are suggested approaches in both diseases, they provide improvement in EVALI patients with some experts comparing response to corticosteroids equivalent to that seen in eosinophilic pneumonia.5,58
Case reports have shown the difficulty of diagnosing patients with EVALI during the COVID-19 pandemic. In these reports, most patients were initially treated for COVID-19 and only after multiple negative nasopharyngeal swabs/reverse transcriptase–polymerase chain reaction tests was EVALI considered.59–61 A recent case series studied seven patients with EVALI, all of whom were seen in clinic/urgent care prior to hospitalization but were undiagnosed.62 Another review studied 12 adolescents who were thought to have COVID-19 but were eventually diagnosed with EVALI.62,63 Thus, it is critical to establish history of vaping products, particularly in young adults, and to maintain a low index of suspicion for EVALI in the appropriate context and during the COVID-19 pandemic.
Conclusion
When assessing patients with respiratory distress with a broad differential, obtaining key history and risk factors, and understanding SARS-CoV-2 test sensitivity are essential for diagnosing EVALI, which remains a diagnosis of exclusion. The psychological manifestations of the COVID-19 pandemic, including social distancing, may have promoted increased vaping tendencies and, thus, increased rates of EVALI. Detecting radiological patterns of acute lung injury and hypersensitivity pneumonitis, identifying vitamin E in BAL samples and vacuolization of macrophages and pneumocytes, and obtaining careful vaping history are important for diagnosing this disease.60,64 The diagnosis of EVALI is of particular importance due to important diagnostic and therapeutic implications in the short and long term. Concerted efforts between physicians and local governing bodies, as well as continued efforts to increase awareness of EVALI among adolescents and young adults are paramount in preventing a further increase in EVALI cases.