Asthma is a common and heterogeneous disease affecting approximately 10% of the population.1 Severe asthma – a subset of the disease that affects a fraction of patients with asthma but comprises more than half of the morbidity, mortality and costs relating to asthma – is predominantly caused by smouldering type-2 eosinophilic airway inflammation.2,3 Until now, the main strategy for tackling this incendiary process and the ensuing bronchial remodelling, asthma attacks and premature deaths has been to douse the fire with oral corticosteroids (OCS).4 The persistence of eosinophilic airway inflammation in some patients with severe asthma indicates that the airway mucosal mechanisms driving the recruitment of eosinophils towards the airway epithelium are or have become resistant to the effects of inhaled corticosteroids. In this situation, OCS are helpful because they deplete the reservoir of circulating eosinophils, thus preventing eosinophil recruitment even in the presence of a strong chemotactic pull towards the airway epithelium.5
While OCS may prevent the inflammation from remodelling airways, they have been shown to reshape the appearance, health status and life of patients.6–9 Consequently, the traditional treatment with OCS contributes to what could be called ‘people remodelling’.
Short and infrequent OCS burst therapies are generally considered safe by physicians; however, they are not.6,8 In both inpatients and outpatients, mood changes and insomnia caused by OCS can lead to disturbing situations for the patients, their family, neighbours and physicians.10 Furthermore, dyspepsia and gastrointestinal ulcers lead to increased healthcare use, while fluid retention is uncomfortable for many patients.6 The frequency and severity of acute and long-term complications related to treatments significantly increase with a lifetime cumulative dose as low as 500–1,000 mg of prednisolone or equivalent.6 Although the treatment is most often necessary, prescribing this treatment implies that we inadvertently start harming patients after two-to-four bursts of OCS in a lifetime.7,8
The long-term impacts of corticosteroid dependence are well recognized. The consequences of even a small daily dose of OCS are, at worst, premature mortality and, at best, the likely possibility of experiencing weight gain and a Cushingoid appearance, as well as an increased risk of osteoporosis and fracture, hypertension, diabetes mellitus, and cardiovascular disorders.6–8 Evidence also suggests that adrenal insufficiency, a potentially life-threatening condition, might affect 60% of OCS-treated patients with asthma.10 Compared with non-corticosteroid-dependent patients with asthma, those treated with OCS have excess incident morbidity of 12 comorbidities per 1,000 patients/year and are estimated to require up to three times more healthcare resources.7,8
The scope, rigour and reproducibility of these toxicological data highlight the importance of making OCS avoidance a shared treatment goal in severe asthma.4 The need for incisive action is especially dire in corticosteroid-dependent asthma. Fortunately, dedicated phase III trials have demonstrated the steroid-sparing effect of three of the six biologics approved for use in asthma.11 They also provide a framework for developing formal weaning protocols.
The first marketed steroid-sparing biologic drug was mepolizumab, an anti-interleukin (IL)-5 monoclonal antibody. In the 2014 SIRIUS trial (Mepolizumab steroid-sparing study in subjects with severe refractory asthma; ClinicalTrials.gov identifier: NCT01691508),12 mepolizumab demonstrated a 50% versus 0% reduction of the median dose of OCS at 24 weeks when compared with placebo, while also reducing the exacerbation rate, improving quality of life and improving lung function. The 4-weekly weaning protocol was initiated 4 weeks after the first injection on the basis of asthma and adrenal insufficiency symptoms.12
The 2017 ZONDA trial demonstrated significant results with benralizumab, an anti-IL-5 alpha-receptor monoclonal antibody (Efficacy and safety study of benralizumab to reduce OCS use in patients with uncontrolled asthma on high dose inhaled corticosteroid plus LABA and chronic OCS therapy; ClinicalTrials.gov Identifier: NCT02075255).13 In this trial, which employed a similar weaning protocol to the SIRIUS trial, the OCS median dose reduction at 28 weeks was 75% with benralizumab versus 25% with placebo. The exacerbation rate was also significantly lower in the treatment group, and symptoms and lung function showed a trend towards improvement.13
More recently, the PONENTE study has held special interest (Study to evaluate efficacy and safety of benralizumab in reducing oral corticosteroid use in adult patients with severe asthma; ClinicalTrials.gov Identifier: NCT03557307).10 The focus of this single-arm study was on rapid OCS tapering in benralizumab-treated patients. The weaning strategy was nearly four times quicker than in ZONDA and SIRIUS, insofar as reductions occurred every 1, 2 or 4 weeks depending on asthma symptoms and programmed adrenal testing. During the 24-week observation period, the median OCS reduction was 100%, and 91% of patients achieved a dose of ≤5 mg prednisolone. Exacerbation rate and asthma control were improved compared with those of the year preceding the study. The positive effect of this large reduction in OCS exposure on people remodelling and other aspects of benralizumab treatment was reflected by a large 1.0-point improvement in the Asthma Control Questionnaire score (minimally important difference: 0.5 points) compared with the score at enrollment.10
Dupilumab, an anti-IL-4 alpha-receptor monoclonal antibody, was also shown to be effective in corticosteroid sparing in the 2018 VENTURE study (Evaluation of dupilumab in patients with severe steroid dependent asthma; ClinicalTrials.gov identifier: NCT02528214).14 Corticosteroid weaning occurred in weeks 4–20 of treatment. Dupilumab-treated patients had a median OCS dose reduction of 100% at 24 weeks compared with a 50% reduction in the placebo group. Exacerbation rate and lung function improved in the biologics group. The quality-of-life evaluation, although showing a tendency towards improvement with treatment, did not meet the clinical endpoint.14 Of these trials, only VENTURE did not require raised type-2 biomarkers for patients to enrol. However, most patients had raised biomarkers (Appendix 1). Therefore, none of the biologic drugs can be assumed with certainty to be corticosteroid sparing in type-2-low asthma.
The ambitious 2022 SOURCE trial evaluated tezepelumab, an anti-thymic stromal lymphopoietin monoclonal antibody (Study to evaluate the efficacy and safety of tezepelumab in reducing oral corticosteroid use in adults with oral corticosteroid dependent asthma; ClinicalTrials.gov identifier: NCT03406078).15 The alarmin anti-thymic stromal lymphopoietin plays a role upstream, where it sets off type-2 and non-type-2 inflammatory pathways. However, blocking this protein did not translate into a statistically significant OCS-sparing effect in the SOURCE trial. Noteworthy methodological differences with the trials mentioned above were the longer weaning period (44 weeks in total) and the possibility of continuing weaning after an asthma attack occurred.15 These differences might have accounted for the differences in outcomes between the SOURCE trial and the other trials, although it is possible that tezepelumab, which acts primarily on the abnormally activated airway epithelium, does not suppress the reservoir of circulating eosinophils enough to be OCS sparing. Nevertheless, significant differences in the OCS-sparing effect of tezepelumab were observed in people with greater baseline blood eosinophil counts. Lastly, omalizumab and reslizumab, the two other biologics approved for severe asthma, have undergone no dedicated corticosteroid-sparing trial.
Across the corticosteroid-weaning trials, adverse events were reported equally between the biologics and placebo groups.10,12–14 Anti-drug antibodies were identified in less than 10% of both patients treated with biologics and with placebo. These antibodies did not seem to have any impact on the treatment’s efficacy during the observation period.
Despite the impressive evidence supporting OCS dose reduction in patients treated with mepolizumab, benralizumab or dupilumab, many challenges still prevent effective dose lowering, even when these biologics are safe and clinically appropriate. The most important obstacle to OCS reduction is patients’ adherence to dose-modification advice. According to Busby et al.,16 up to 30% of patients did not reduce their OCS doses when advised to do so, even though they voluntarily and knowingly enrolled in an algorithm-driven reduction trial. Strikingly, the adherence levels were largely dependent on the recruiting centre, suggesting that the local culture and the manner in which healthcare professionals present the advice are important factors.16 This may translate into even lower adherence to advice in a real-world clinical setting.
In practice, it is difficult for clinicians to taper OCS treatments of patients dependent on corticosteroids without prespecified trajectories. To our knowledge, few institutions use a standardized protocol to systematically enact these changes or provide resources for patients who are understandably worried about stepping down from a previously life-saving salvage treatment. Inspired by the SIRIUS, ZONDA, VENTURE and PONENTE trial protocols10,12–14 and by pharmacodynamic data indicating that benralizumab acts immediately,17 we herein propose an agent- and patient-dependent protocol for corticosteroid weaning when initiating a biologic treatment (Figure 1).
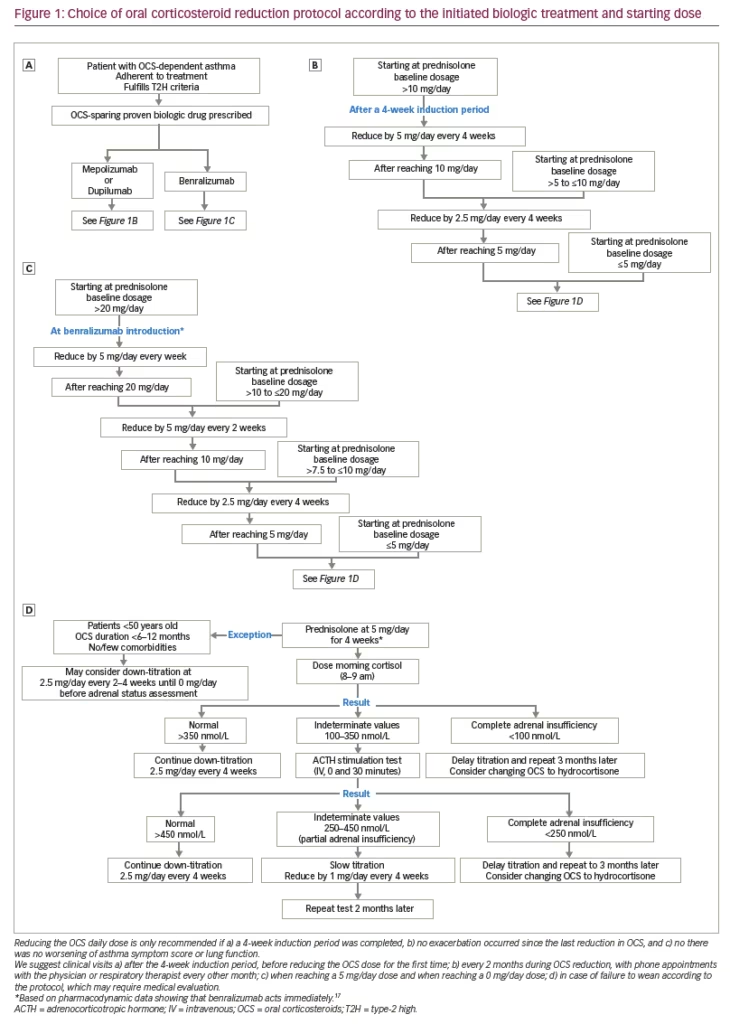
Conclusion
In conclusion, although life-saving, corticosteroids induce a debilitating process of ‘people remodelling’. Reducing OCS while maintaining asthma control is possible with mepolizumab, benralizumab and dupilumab. However, many challenges still stand in the way of corticosteroid reduction by clinicians. The proposed personalized corticosteroid weaning protocol (Figure 1) is intended to alleviate some of these obstacles.