Respiratory Function Loss and Respiratory Endpoints in DMD
Duchenne muscular dystrophy (DMD) is the most common and devastating type of muscular dystrophy. Lack of the protein dystrophin causes severe and progressive myofibre degeneration, general muscle weakness and wasting. With increasing age, DMD patients are confronted with loss of ambulation, loss of upper limb function, cardiac dysfunction and dependence on mechanical airway clearance and mechanical assisted ventilation representing irreversible and lifechanging events of disease progression. Although early diagnosis and multi-stage disease management regimes (e.g. Bushby et al.)1,2 increase quality of life and life expectancy, the disease is still associated with early morbidity and mortality. In DMD, progressive weakness of the chest wall muscles precedes weakness of the diaphragm (used predominantly for inspiratory function) and leads to restrictive lung volume changes measured as reduced total lung capacity and forced vital capacity (FVC).3–7 Initially, this loss of lung volume results from the inability to pull up the respiratory system to total lung capacity and to push it down to residual volume. In later disease stages, additional restrictions occur as a result of progressing muscle fibrosis and changes in lung and chest wall recoil, thoracic wall compliance and spinal deformities (i.e. scoliosis).
In the late first decade the earliest signs of respiratory impairment manifest by reduced static airway pressures (maximal expiratory and inspiratory pressures). The gradual loss of respiratory function in DMD measured by spirometry usually begins early in the second decade and progresses to restrictive pulmonary syndrome, impaired respiratory secretion clearance, life-threatening pulmonary infections due to ineffective cough, nocturnal and daytime hypoventilation, obstructive apnoeas and eventually respiratory failure during the late second or third decade of life.3,8–10
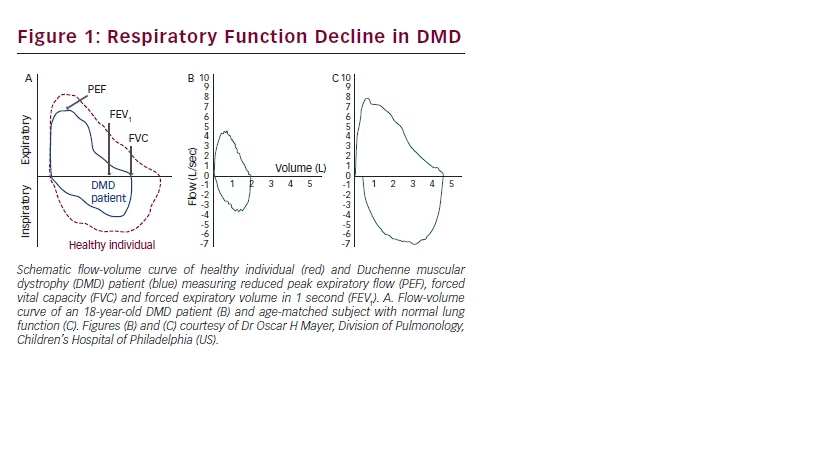
In the absence of obstructive pulmonary disease, FVC (measured in litres), peak expiratory flow (PEF, measured in l/minute), and forced expiratory volume in 1 second (FEV1, measured in litres) are major interrelated spirometry measures reflecting both inspiratory and expiratory muscle force impairment and restrictive lung volume changes due to neuromuscular weakness. In DMD both inspiratory and expiratory respiratory weakness is indicated by abnormal flow-volume curves (see Figure 1).
FVC is used to assess respiratory muscle involvement in many neuromuscular diseases, such as DMD. One may infer the presence of a restrictive ventilatory defect due to neuromuscular weakness when the FVC is reduced and the FEV1/FVC ratio is normal or increased. Previous studies have shown excellent test–retest reliability of FVC among DMD subjects. However, a limitation of FVC as a respiratory endpoint in DMD is that it is potentially affected by thoracic wall compliance/ fibrosis and thoracic deformities resulting from progressive scoliosis. FEV1 typically follows the decline measured in FVC. In the absence of obstructive pulmonary disease such as asthma, FEV1 is also an indicator of respiratory impairment due to neuromuscular weakness of combined inspiratory and expiratory weakness.
In DMD patients who do not exhibit bronchial obstruction, PEF reflects expiratory muscle force.5 Abnormal respiratory mechanics in DMD are not limited to the lung and chest wall and may also involve the upper airways.6 Therefore, respiratory strength in DMD (assessed by PEF) is a measure not only of expiratory strength but also inspiratory effort and upper airway resistance, which are both abnormal in DMD.11,12 There is a theoretical possibility that PEF may be more sensitive to a treatment intervention than FVC due to the impact of fibrosis and chest wall deformities on FVC. All three of these measures – PEF, FVC and FEV1 – can be obtained with high reliability in DMD patients older than ~8 years.
As respiratory function tests are influenced by body growth and age, these measures are typically normalised to height-matched (PEF)13 or height- and age-matched (FVC and FEV1)14 normative populations and expressed as ‘per cent predicted’ values (PEF%p, FVC%p, FEV1%p).
Recent care guidelines recommend changes in DMD disease management as soon as patients fall below certain thresholds in FVC.2,15–17 For example, preoperative training prior to surgical procedures and post-operative use of non-invasive ventilation should be strongly considered for patients with FVC <50 %p, and is necessary for patients with FVC <30 %p. Various levels of impairment of FVC have been reported to be prognostically associated with an increased risk of respiratory complications and death in DMD.15,18
A similar method of categorising the severity of lung function impairment based on the FEV1%p values has been described.19 These severity scores can be associated with performance, such as ability to work and function in daily life, morbidity and prognosis.20–23 A drop of FEV1%p to below 40 % was found to be a sensitive indicator of sleep hypoventilation in DMD.24 Thus, reduction in the rate of progression of FEV1%p would be of direct clinical value to a patient with DMD in terms of slowing pulmonary disease progression and decreasing the likelihood of developing carbon dioxide retention.
More recently, peak cough flow (PCF) measures are obtained as part of standard of care. When PCF falls below 160 l/minute, cough is no longer effective enough to provide adequate mucociliary clearance.25–27 According to published standard of care considerations, manual and mechanically assisted cough techniques are necessary when either respiratory infection is present and PCF is 270 l/minute, or in the absence of a respiratory infection when PCF is 160 l/minute. One of the challenges in PCF measures tends to be reduced reliability of testing in children and the difficulty of performing the test.25
In summary, PEF, FVC and FEV11 are three inter-related pulmonary parameters and reflect both inspiratory and expiratory muscle force impairment, and restrictive lung volume changes indicated by abnormal flow-volume curves in DMD. The %-predicted values control for maturational changes longitudinally. PEF%p, FVC%p and FEV1%p are also highly correlated in DMD and are all indicators of restrictive pulmonary disease due to neuromuscular weakness and are the most reliably assessed respiratory measures in this disease. Finally, natural history studies show that these are the only three respiratory function endpoints with consistent declines across the second decade in DMD from age 10–18 years.28,29
Glucocorticoid Steroids – A Delicate Benefit–Risk Balance
There is agreement that patients with DMD generally benefit from glucocorticoid treatment,30 the therapeutic objective of which is to slow the decline in muscle strength and stabilise respiratory function. The effect of glucocorticoids on muscle strength has been shown to prolong ambulation31,32 and continued treatment after the patient becomes nonambulatory has shown reduction in the risk of progressive scoliosis and stabilisation of respiratory test variables.33,34 These and the results of other clinical trials support the use of glucocorticoids,35 which have become the mainstay of disease management in DMD.1,2
However, the mode of action of steroids in DMD still is not entirely resolved and their chronic administration is associated with well-described significant risks, such as growth retardation, bone demineralisation and increased fracture risk, obesity, insulin resistance and hyperglycaemia, cutaneaous complications (acne, striae), arterial hypertension, cardiomyopathy, cataracts and important behavioral changes, such as increased anxiety and anger,1,36 which especially after the loss of ambulation, can negatively affect the benefit–risk balance of glucocorticoid use. Furthermore, while there are emerging data concerning benefits of long-term administration of steroids for preservation of upper limb and respiratory function,32,37 the effectiveness and risks of specific glucocorticoid treatment regiments in preventing scoliosis, maintaining upper limb function and stabilising respiratory function in the older non-ambulant patients with DMD has not been studied in
controlled trials. At any time, approximately 40 % of DMD patients are either glucocorticoid-naïve or have discontinued treatment,32 due to glucocorticoid side effects and partial treatment effect, and this proportion even increases in the later post-ambulatory phase. Therefore, many patients are left without pharmacological treatment options at around the age when patients become non-ambulatory and the decline in respiratory function becomes clinically relevant. In summary, there is an unmet medical need for the management of respiratory function deterioration in DMD patients not taking glucocorticoids or in patients who do not respond to glucocorticoid treatment.
Rationale for Idebenone as Treatment Approach for DMD
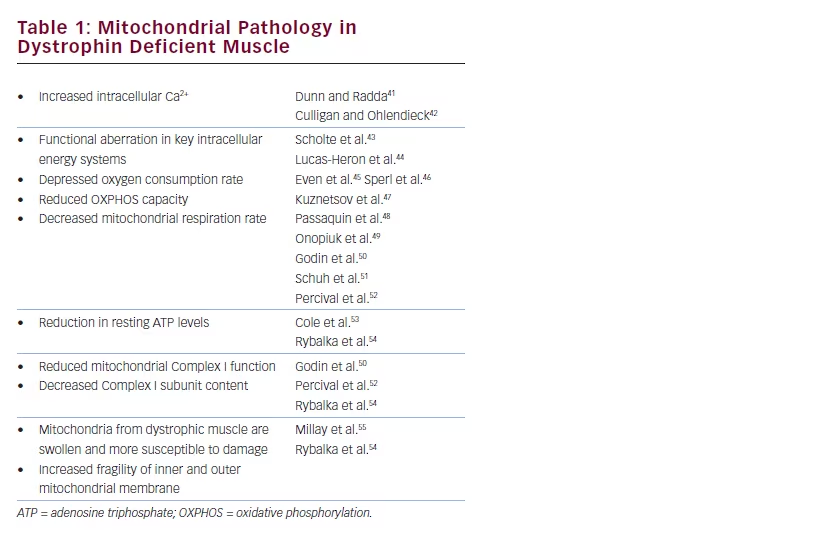
There is an increasing body of literature (see Table 1)41–55 indicating that dystrophin deficiency causes sarcolemmal fragility and intracellular Ca2+ dysregulation, which generally leads to mitochondrial dysfunction, a reduction in resting adenosine triphosphate (ATP) levels, an increased production of cell-damaging reactive oxygen species (ROS) and ultimately mitochondrial damage.38–40
The combination of primary dystrophin deficiency and secondary mitochondrial dysfunction and impaired muscle cell regeneration, degeneration and cell death is shown in Figure 2A. Muscle membrane injury caused by dystrophin deficiency leads to increased calcium influx and intracellular calcium accumulation. Thus, the primary pathogenesis results in increased ROS and damage to and dysfunction of mitochondria with subsequent depletion of mitochondria over time.
Recently, Complex I function was shown to be deficient in mitochondria from the mdx mouse and ATP production was ameliorated by stimulating Complex II.54 As direct evidence of a mitochondrial role in the pathogenesis of muscular dystrophy, mitochondrial biogenesis has been demonstrated to ameliorate the pathology and improve function in the dystrophin deficient mdx mouse model. Specifically, Selsby and colleagues56 showed that over-expression of peroxisome-proliferator receptor γ coactivator 1α (PGC-1α), resulted in increased utrophin and slow type I myosin heavy chain fibre expression, as well as elevated mitochondrial protein expression and increased expression of oxidative genes. These PGC-1α over-expressing mdx limb muscles were better able to maintain force during eccentric lengthening contractions compared with control limbs and demonstrated reduced disease-related muscle injury as measured by decreased areas of necrotic fibres and centrally nucleated fibres. In addition, it has long been known that mitochondrial depletion is associated with greater disease progression in DMD.57,58
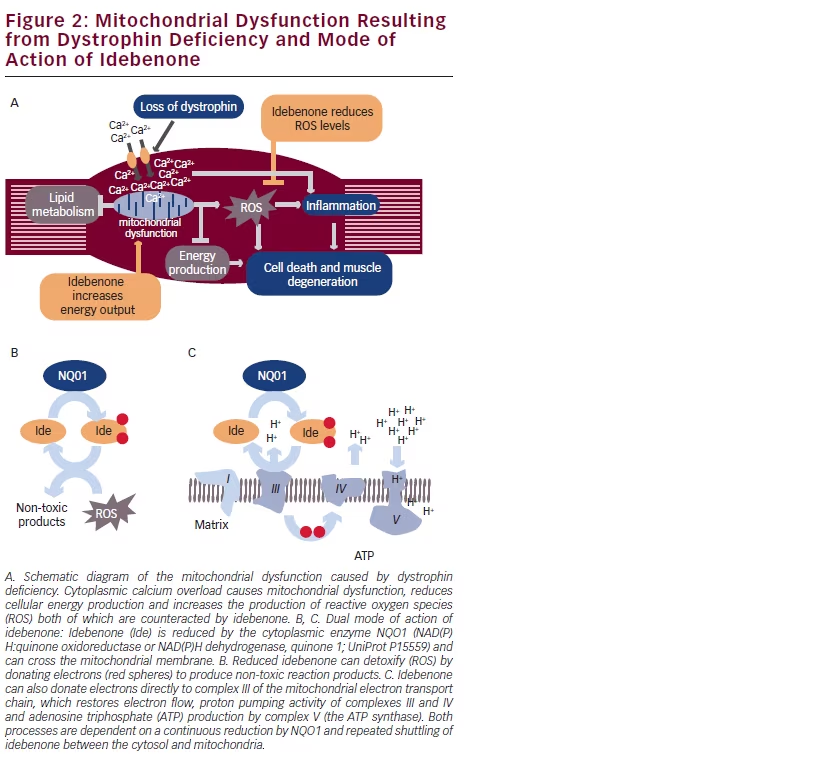
Based on these findings, mitochondrial activation via stimulation of Complex II- or Complex III-mediated electron flux might be a therapeutic approach to overcome mitochondrial dysfunction in dystrophindeficient muscle.
Idebenone is a synthetic short-chain benzoquinone, chemically derived but clearly distinct from CoQ10 in its subcellular distribution and pharmacological action due to its shorter carbon-tail and terminal hydroxyl group resulting in higher hydrophilicity and the potential to cross membranes.59,60 Idebenone has been reported to utilise and activate Complex I-independent metabolic pathways61–64 by the transfer of energy equivalents from the cytosol directly into the mitochondrial electron transport chain (ETC). Upon entering the cell, idebenone is efficiently reduced by the cytoplasmic enzyme NADH-quinone oxidoreductase 1 (NQO1) and the resulting reduced form of idebenone
enters the mitochondria where it transfers its electrons to Complex III. This mechanism thereby bypasses complex I-dependent electron flow and as a result is able to produce ATP even in situations where Complex I is dysfunctional.60,62,63 Separately, idebenone has been reported to be a powerful inhibitor of ROS formation.61,65,66 The mode of action of idebenone is illustrated in Figure 2B, C.
Idebenone significantly increased the daily running distance of mdx mice and reduced cardiac inflammation and fibrosis in a long-term blinded placebo-controlled study.67 Idebenone also ameliorated cardiac diastolic dysfunction and reduced mortality from systolic cardiac pump failure in this model. Considering the generally accepted limitation of the mdx mouse as an animal model for DMD, these potential benefits on skeletal muscle degeneration, endurance, and cardiac function would need to be confirmed in patients with DMD.
Clinical Development of Idebenone as a Treatment Option for DMD
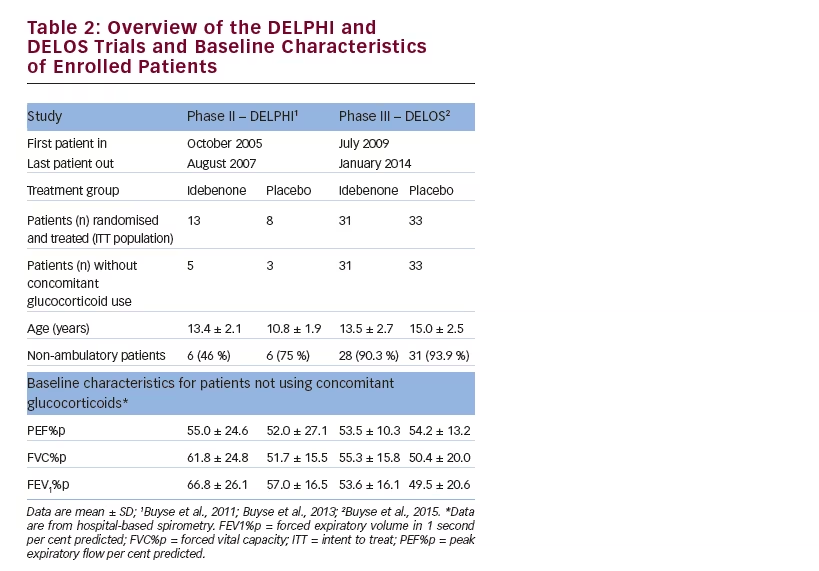
Two randomised, placebo-controlled clinical trials were conducted to assess the efficacy and safety of idebenone on respiratory function outcomes in DMD (see Table 2). DELPHI was a phase II, single-centre study,68 while DELOS was an international, multicentre phase III study.69
The exploratory phase II DELPHI study reported a better outcome on PEF%p in the idebenone-treated group compared with the placebo group when all patients (irrespective of their concomitant glucocorticoid use) were analysed.68 However, in a post-hoc analysis is was found that the treatment effect of idebenone on respiratory function outcomes was considerably larger in patients not using concomitant glucocorticoids.70 This could be explained by the protective effect of glucocorticoids, challenging a formal demonstration of an additive effect on top of steroids within the limitations of a human clinical trial. Although the number of patients in the subgroup of patients not using glucocorticoids was too small to allow a firm conclusion, the expected influence of concomitant glucocorticoid use was considered in the planning of the confirmatory phase III DELOS study.
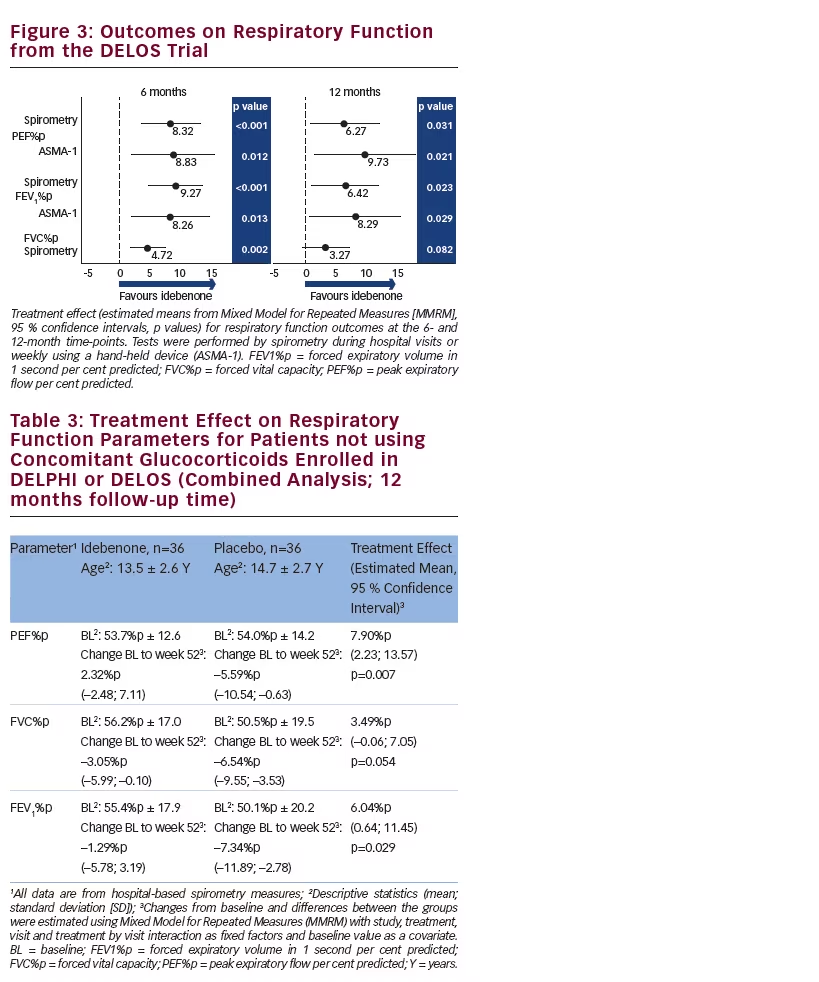
The study objective of DELOS was to test whether idebenone could slow the decline in respiratory function in patients with DMD not taking concomitant glucocorticoids.69 The 64 patients (age 10–18 years) randomised to idebenone (n=31) or placebo (n=33) treatment had washed out from previous glucocorticoid use prior to enrolment and did not use glucocorticoids for the treatment of the underlying disease during the 12-month follow-up period. The study met its primary endpoint demonstrating a statistically significant and clinically relevant treatment effect on PEF%p from baseline to month 12, which was supported by the consistency in results obtained for lung volume parameters (FVC%p, FEV1%p) at the 6- and 12-month time-points measured by hospital-based spirometry or at home using a hand-held ASMA-1 device (see Figure 3).
Published natural history data confirm that PEF%p, FVC%p and FEV1%p all show a linear decline from age 10 onwards.28,29 The treatment effect of idebenone reported in DELOS for patients 10 years and older not using concomitant glucocorticoids on PEF%p, FVC%p and FEV1%p is equivalent to slowing respiratory decline by at least 1 year, which is clinically meaningful.
The findings on respiratory function parameters in the DELOS study were supported by additional clinical observations. Specifically, the proportion of patients with reductions in FVC or PCF below clinically important thresholds, known to be predictive of imminent ventilatory failure, and the reduced number of upper and lower airway tract related disease in the idebenone group, are strongly supportive for the clinical meaningfulness of the idebenone effect.69
In order to further assess the efficacy of idebenone in delaying the loss of respiratory function, data from glucocorticoid non-using patients enrolled in DELPHI and DELOS were combined for analysis (see Table 3). The combined analysis of all patients enrolled in DELPHI or DELOS demonstrated a significant decline in PEF%p from baseline to month 12 in the placebo group compared with a slight improvement in the idebenone group, resulting in a significant difference of 7.90%p in PEF%p between treatment groups. Similar combined analyses for FVC%p and FEV1%p revealed consistent treatment effects in favour of idebenone.
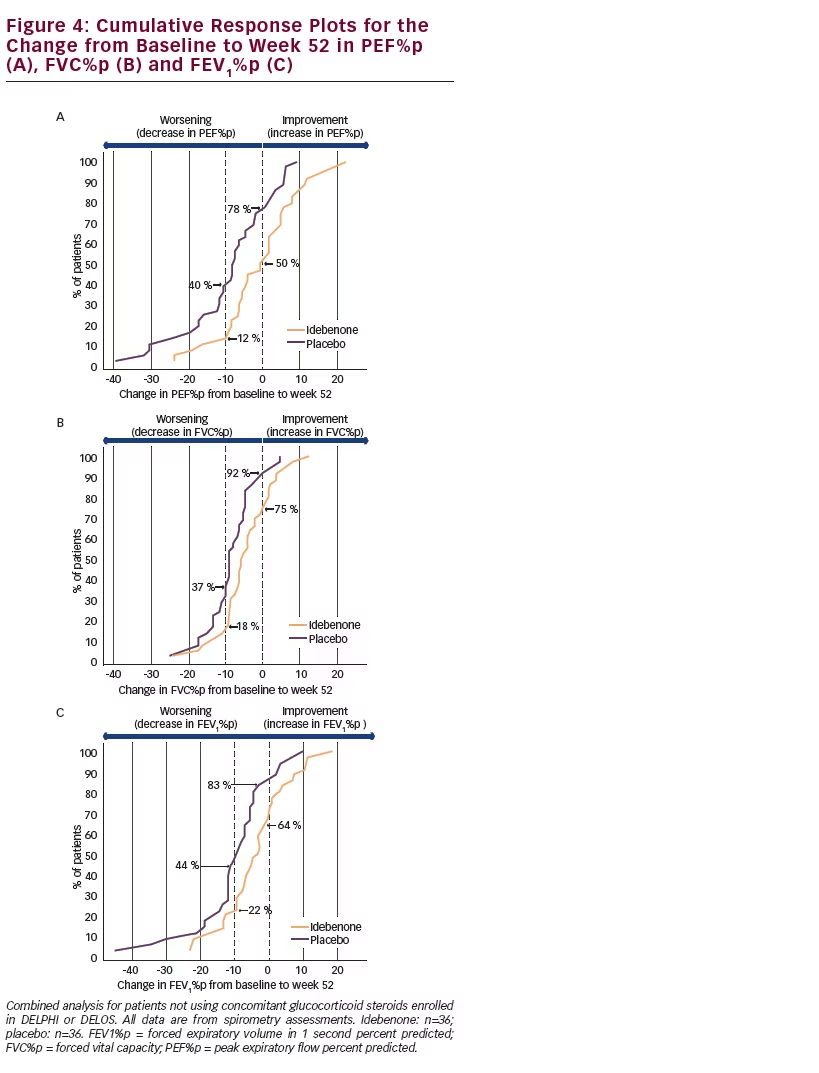
The consistency and robustness of the findings for the combined analyses is also demonstrated in cumulative response plots where the change from baseline to the 12 month (52 week) time-point is displayed for each patient (see Figure 4). For all three respiratory function parameters there is a clear separation of the curves for idebenone-treated patients from those in the placebo group. For PEF%p (see Figure 4A) there were fewer patients in the idebenone group who declined compared with placebo (50 % of patients in the idebenone group compared to 78 % in the placebo group declined in PEF%p). Likewise there were fewer patients in the idebenone group who declined in PEF%p by 10%p or more compared with the placebo group (12 % of patients in the idebenone group compared with 40 % in the placebo group). Similar patterns in favour of idebenone are also seen for FVC%p and FEV1%p (see Figure 4B, C).
Idebenone in the Emerging Landscape of DMD Treatment Options
Although best practice guidelines on their dosing regimen are still lacking, glucocorticoids have beneficial effects and can slow disease progression particularly in younger, still ambulatory patients with DMD (e.g. Bushby et al.)1,2 However, not all patients benefit to the same extent from glucocorticoids and their benefit–risk profile in older, non-ambulatory patients still needs to be established. Therapeutic approaches to partially circumvent the effect of nonsense-mutations in the dystrophin gene or to partially correct dystrophin expression by exon skipping approaches are in advanced clinical investigation. So far, it appears that their therapeutic potential is best deployed when applied in younger, ambulatory patients with the goal to preserve muscle force and function. However, therapeutic options for older, non-ambulatory patients not able to use glucocorticoids are currently lacking, which comprises approximately 40 % of the entire DMD patient population 10 years and older.32 Based on the data from the randomised placebo-controlled studies summarised above, idebenone can be considered a suitable extension of the emerging landscape of treatment options for DMD. In DELOS, the firstever phase III trial in DMD with a positive outcome, significant and clinically relevant results for primary and secondary endpoints coherently showed that idebenone reduced the loss of respiratory function. Results from the DELOS study indicate that patients who previously used glucocorticoids for the treatment of the underlying disease but did no longer tolerate their use benefit from idebenone to the same extent as patients who never took glucocorticoids.69 This is an important observation as it shows that glucocorticoids and idebenone could be used sequentially in the same patient. Although data from the current trials were obtained primarily in patients not using glucocorticoids, there is no reason a priori why idebenone could not also be exerting a treatment effect in patients using glucocorticoids concomitantly. It remains to be seen whether longer-term administration of idebenone will show incremental benefits over glucocorticoid treatment with regard to stabilisation of pulmonary function. Likewise, in theory, idebenone treatment might be combined with different therapeutic approaches such as exon skipping or stop-codon read-through. However, at the current time it is premature to speculate about its possible use in conjunction with any such therapy approaches.
Conclusion
Improved patient care with best-practice recommendations and the introduction of glucocorticoids has increased the quality of life and survival time of patients with DMD. Nevertheless, with increasing age loss of respiratory function continues to be a predominant cause of early morbidity and mortality. Efficacy data from randomised placebocontrolled phase II and III trials show that idebenone significantly reduced the loss of respiratory function in 8–18 year old patients who were not taking concomitant glucocorticoids. Considering its favourable safety and tolerability profile, this oral medication could therefore become the first treatment option for patients not using glucocorticoids with the possibility to ameliorate a life-threatening complication in the ageing population of DMD patients.