Case Presentation
The patient described was diagnosed with cystic fibrosis (CF) at 2 years of age. She was pancreatic insufficient and had an F508del homozygous genotype. Her pretransplant CF-course was complicated by CF-related diabetes, chronic sino-pulmonary colonization with Pseudomonas aeruginosa (but not Aspergillus), CF-liver disease with stable cirrhosis and portal hypertension and normal synthetic function, osteoporosis, and a central line-associated venous thromboembolism. She received a lifesaving double-lung transplant in her late thirties.
Her postoperative course was initially uncomplicated, with surveillance bronchoscopies following the transplant failing to identify pathogens including fungi. No postoperative antifungal prophylaxis was received. Post-transplant medications include prednisone 5 mg orally once daily, azathioprine 100 mg orally twice daily, and tacrolimus to a target trough level of 8–10 ng/mL. Allergies include a biopsy-proven mycophenolate mofetil-induced colitis and ceftazidime rash.
The first pleural-based issues began 10 years following transplant. By this time she had progressed to have mild chronic renal failure with a glomerular filtration rate (GFR) of 52 mL/min/1.73m2. She presented with fever and dyspnea, and was diagnosed with a right lower lobe pneumonia, which included a progressive parapneumonic effusion. She recovered to baseline after a course of cefepime and metronidazole. Sputum cultures grew the same pre-transplant P. aeruginosa strain, and were negative for fungi and mycobacteria. Pleural fluid analysis revealed an exudative effusion (pleural fluid: white blood cells [WBC] 0.6 x 109/L, pH 7.42, lactate dehydrogenase [LDH] 189 U/L, total protein 36 g/L, glucose 5 mmol/L; serum LDH 125 U/L, serum total protein 74 g/L), but all cultures were negative. A small pleural collection persisted.
Over the subsequent year, the patient experienced progressive fatigue and dyspnea on exertion with no constitutional symptoms. Serial radiographs and computed tomography (CT) scans revealed no change to the small pleural collection. She received a course of intravenous iron sucrose and darbepoetin alpha for an iron deficiency anemia with no significant improvement in hemoglobin.
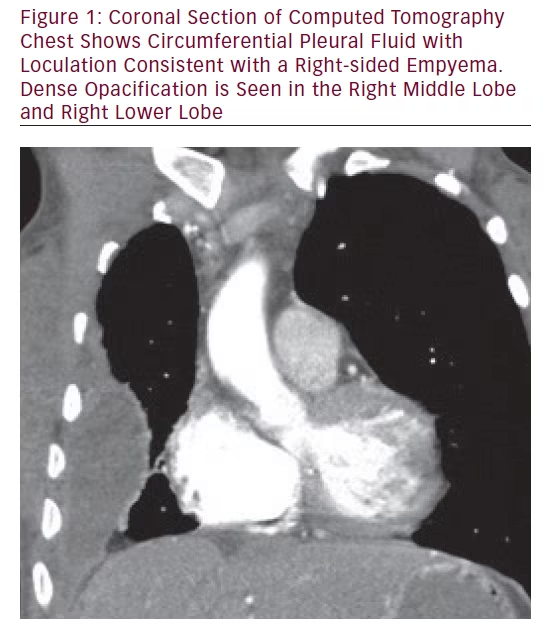
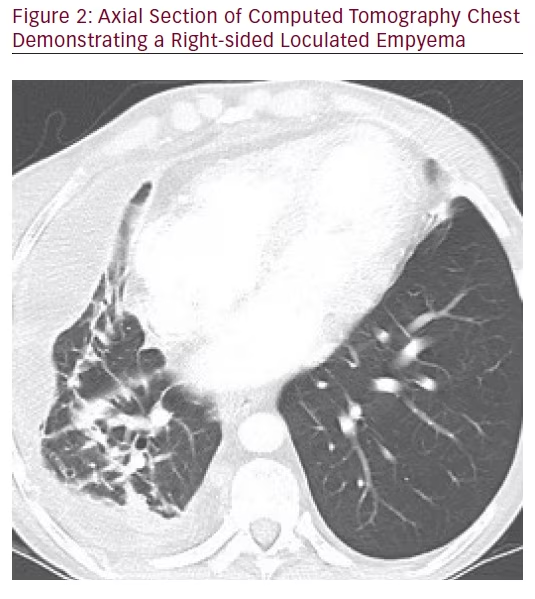
One year later she presented with subacute worsening fatigue, but no fevers or change in cough. Her hemoglobin was 77 g/L, platelets (PLT) 423 x 109/L, WBC 9.2 x 109/L, C-reactive protein (CRP) 107.5 mg/L. Chest X-ray showed an increase in the pleural effusion in the right lower lobe. Chest CT images are shown in Figures 1 and 2. Repeat thoracentesis prior to receipt of antibacterials revealed an exudative pleural effusion: pleural fluid WBC 153 x 109/L, LD >1,800 U/L, total protein 43 g/L; serum LDH 134 U/L, serum total protein 73 g/L. Aspergillus fumigatus grew from the pleural fluid cultures (with the following antifungal minimal inhibitory concentrations reported: voriconazole 1 μg/mL, micafungin 0.015 μg/mL, and amphotericin B 1 B 1 μg/mL). Meropenem plus levofloxacin had been initiated empirically after thoracentesis, but were discontinued when cultures were finalized as Aspergillus fumigatus. Antifungal therapy was started within 8 hours of the thoracentesis with voriconazole (6 mg/kg
intravenous [IV] load for two doses followed by 4 mg/kg every 12 hours) plus micafungin (100 mg IV every 24 hours) with the recognition of fungal hyphae in pleural fluid. Notably she lacked significant risk factors predisposing to invasive aspergillosis (IA). Six days after the recognition of fungal empyema, an attempt at videoassisted thoracoscopic surgery (VATS) was performed. Appropriate surgical planes could not be visualized; therefore, an open right thoracotomy and decortication was performed. The lung was completely mobilized and the pleural-based disease around the anterior and lower aspect of the chest wall was removed. Operative cultures from the pleural fluid, pleural tissue, and chest wall grew A. fumigatus despite therapy, with no other organisms isolated. Two chest tubes were inserted intraoperatively for drainage.
She developed progressive hepato-renal dysfunction 5 days following surgery manifesting a GFR of 13 mL/min/1.73m2 and profound hyperbilirubinemia. Prior to this, voriconazole trough concentrations were measured on two occasions, and both were within therapeutic range (2.2 mg/L and 3.2 mg/L).1 Voriconazole was discontinued and she was changed to a salvage antifungal regimen of caspofungin.2 Unfortunately, due to progressive deconditioning, unremitting hepatic encephalopathy, and acute on chronic kidney injury, she expired 28 days following the identification of her fungal empyema necessitans with her family at her side.
Discussion Empyema necessitans was first reviewed by Sindel in 1940, with 81 cases due to Mycobacterium tuberculosis, and 31 by primarily Streptococcus pneumoniae.3 Two cases of empyema necessitans secondary to Pseudomonas cepacia (Burkholderia cepacia complex) were reported post double-lung transplant for CF.4 To our knowledge, this is the first case of empyema necessitans due to A. fumigatus in a CF lung-transplant recipient. Also unique is the late onset of pleural disease in our patient—a decade after her initial transplant. Empyema is not an infrequent complication early after lung transplant,5 but the incidence of empyema long after transplant has not been well described.
In 100 cases of lung transplant recipients post-transplantation, Ferrer et al. found seven cases of death all caused by Aspergillus as the etiologic organism, including two cases of Aspergillus empyema,6 highlighting the high morbidity and mortality of this disease. Fungal empyema is difficult to treat and often requires surgical management for possible decortication. It is associated with a reported mortality of 73 %, although this included patients who died without antifungal therapy, prior to knowledge of culture results.7 Those receiving surgical interventions appear to have lower mortality.7
Risk for IA is greatest in the first year following transplant and specific risk factors include: airway colonization with Aspergillus species prior to or following transplant, anastamotic ischemia, bronchiolitis obliterans syndrome, increasing immune suppression (particularly with the use of high-dose steroids and anti-lymphocyte therapies) and concurrent cytomegalovirus (CMV) infection or hypogammaglobulinemia.8–11 Prophylaxis against IA is a contentious matter. Targeted prophylaxis in those at the greatest risk for disease may be the most effective, least toxic, and financially prudent means of preventing IA.8 However, the use of antifungal prophylaxis in those without prior Aspergillus colonization has not been shown to be protective against invasive infection and has been associated with a marked increased risk for squamous cell cancer. Accordingly prudent use of azole-based antifungal prophylaxis is required.12
Mortality of IA in solid organ transplant cases has been reported at 35 %.13 Hepatic insufficiency has been identified as an independent poor prognostic indicator in solid organ transplant patients with IA. This patient did not die directly due to her invasive fungal infection, but rather from complications of multi-organ dysfunction. With multiple comorbidities including pre-existing cirrhosis, she was at an increased risk for postoperative complications. Chen et al. reported an overall in-hospital mortality rate of 30 % in 63 cirrhotic patients with bacterial empyema treated with thoracoscopy, thoracentesis, or tube thoracostomy.14 This patient’s unfortunate scenario of aborted thoracoscopy worsened her chances of survival, as she was subject to the higher risk open thoracotomy/decortication procedure. The patient had a Child-Pugh score of 7 at the time of operation (Class B)—total bilirubin 15 μmol/L, albumin 18 g/L, no ascites, international normalized ratio (INR) 1.3, no encephalopathy—but quickly progressed to a Child-Pugh Class C immediately postoperatively. Child-Pugh Class C is an independent risk factor for in-hospital mortality.14
Medical management of fungal empyema necessitans is limited by the toxicity of available antifungals and pleural drug penetration. The current guideline for IA recommends voriconazole as the primary first-line agent, with multiple alternative or salvage agents.2 However, Marr et al. recently published a large trial of immunocompromised patients with IA, comparing voriconazole plus placebo versus voriconazole plus anidulafungin. Results favored combination therapy, although statistical significance was not attained.15 Initial therapy with an amphotericin B preparation has been associated with increased risk for death in addition to intrinsic nephrotoxic potential.7 In this case, due to the severe burden of disease, combination therapy was started initially. We used voriconazole in combination with micafungin (anidulafungin and caspofungin are not routinely available at our institution) at the time point of identifying fungal elements in the patient’s pleural fluid. Indeed, both voriconazole and micafungin have been shown to penetrate well into the pleural space,16 whereas penetration of liposomal amphotericin B in pleural fluid is relatively low.17. Unfortunately, unremitting hepatic dysfunction precluded ongoing therapy with a triazole, and necessitated a change of therapy where caspofungin was organized owing to its documented role in salvage therapy.2
Conclusion
Aspergillus fumigatus empyema necessitans is a rare and potentially lethal complication, which may present many years after lung transplant. The authors advocate an aggressive medical and surgical approach, as well as consideration for combination therapy with multiple antifungal agents. However, even with aggressive therapy morbidity and mortality remains high, and outcomes may be heavily influenced by the patient”s pre-existing comorbidities.